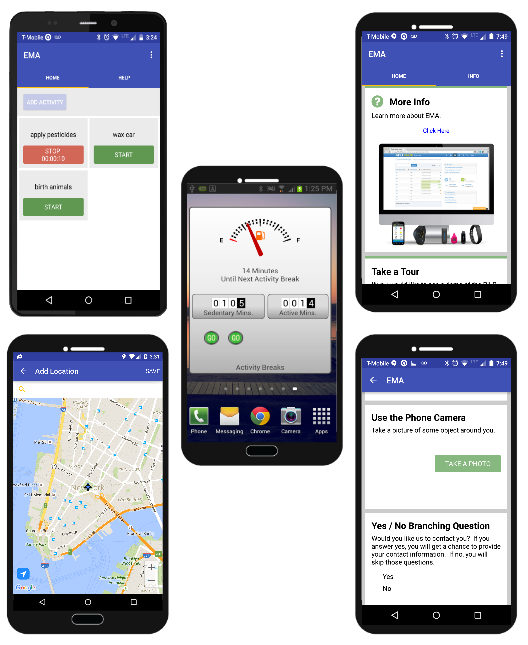
PiLR EMA Mobile App Features
PiLR EMA Product Capabilities
PiLR EMA is a mobile app that is available in Apple iTunes and the Android Playstore. It is written by the same team that built PiLR Health, and is designed with the experience gained from 4 years of custom applications built to meet the needs of researchers. With PiLR EMA and PiLR Health you can:
- Create deploy a complex mobile app to collect data and render content without programming using our EMA Builder
- Write your own simple or complex survey
- Configure rules for translating sensor activity into counts, or activity types (walking, running)
- Set triggers and conditions for when to present a survey to a participant
- Run in background and foreground modes. In background mode, PiLR EMA can implement complex rules to determine when to present a study participant with a survey
- PiLR EMA can access the phone sensors, so it is capable of evaluating rules that use GPS location, activity levels, time and date.
- Run normally even when there is no network connection. Plus, PiLR EMA caches data and forwards it when a network connection is available (wifi or cellular networks)
- Gather data from or deliver configurations to bluetooth sensors
- The mobile app can accept new rules from PiLR Health, so participants’ experiences can change while the study is active
PiLR EMA Feature List
- Support for multiple languages, character sets and graphics. Content and tab labels can be written in any language and can include “emoji”. Buttons and other navigation items already appear in English, Chinese, Spanish, French or Japanese (with other languages being added).
- Support for multiple EMA configurations – allowing content and experiences to be differentiated by cohort and/or by time period
- Engagement features
- Identical experience between iOS and Android
- Capture data from sensors on a smartphone
- Location
- Accelerometer
- Gyroscope
- Sound
- Image/video
- Survey constructions
- Presentation cards of many types, including
- Instruction
- Informational: text, images and/or links
- Survey Queue (entry point for surveys)
- Personal venue (allowing user to mark and name a location)
- Compliance feedback (e.g., score or $ for activities)
- Video, image and audio
- Input cards
- Text
- Select (single/multiple choice, Likert scale)
- Numeric
- Media – to submit audio, video or image
- Triggers in real time
- Date / time of day, optionally with random window
- Location by personal venue and complex geofencing with plot events
- Proximity
- Motion / physical activity (from mobile phone or wearable)
- Survey submitted
- Process controls
- Branching
- “Piping” of answer from one question to a later question
- Conditionals, such as time and count limits
- Do not disturb time
- Presentation cards of many types, including
- Location and path logging
- Notifications and reminders scheduled up to 1 year in advance
- Anonymous app delivery and account registration through a QR code or link
- Automated patient recruitment, inclusion survey, consent and study registration
- Dyad (e.g. parent/child or caregiver/patient) configuration
- Flash cards for cognitive tests
- Interface to Nutritionix food database
- Interface to MOVES app data
- Real time interface to external sensors such as Actigraph
- “Life in a Day” configurations for task and time tracking tied to follow up surveys
- Options to cache all data on the mobile device or delete as being uploaded (for added security)
- Apply in-app password for privacy if the mobile device is shared
- Option to exchange data only over WiFi (to conserve cellular data)